
As six tubes are used, the final reduction of germs/cells will be 10 -6 (1 in 1,000,000). Nevertheless, the importance of locally optimal designs is in that they provide a.Repeat the same process with the remaining test tube, taking 1 ml from the previous tube and adding to the next 9 ml diluents.The second tube now has a total dilution factor of 10 -2. amount of either the original or diluted sample to a tube of melted agar, mixing the tube and pouring the mixture into.Then dilute it and pour into a second test tube.Now, take 1 ml of mixture from the 10 -1 concentration test tube.Discharge a tip of pipette, and insert a new pipe tip into the pipette.Dilution is thoroughly mixed by removing and filling the pipette several times.

This provides an initial dilution of 10 -1. Then add sample solution to the first tube to form a total volume of 10 ml.Serial Dilution Besides the more conventional uses described above, serial dilution may also be used to reduce the concentration of microscopic organisms or cells in a sample.
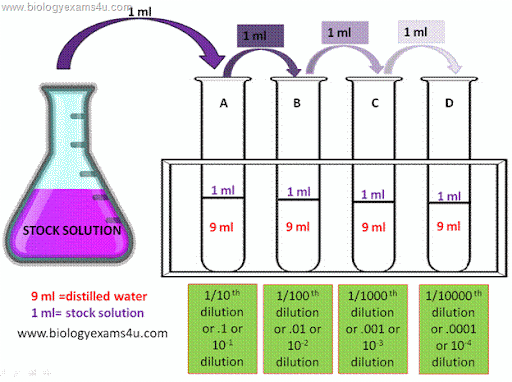
A certain amount of bacteria are eliminated with. Serial dilutions are used to accurately create highly diluted solutions as well as solutions for experiments resulting in concentration curves with a logarithmic scale. It also used to avoid having to pipette very small volumes (1-10 l) to make a dilution of a solution.
THE IMPORTANCE OF SERIAL DILUTION SERIES
After that, a small average volume of each purifier is used to make a series of pouring or dispersing plates.Serial dilution involves the process of taking a sample and diluting it through a series of standard volumes of sterile diluent, which can be either distilled water or 0.9 % saline solution.
